Mitochondrial toxicity in drug discovery
Drug-induced mitochondrial dysfunction is linked to compound attrition, organ toxicity, black-box warnings, and market withdrawals. Identifying mitochondrial toxicity during the early stages of drug discovery therefore has huge cost benefits.
Mitochondrial function is typically probed using end-point assays such as the glu/gal assay, but often or not these assays lack precision and sensitivity, leading to late-stage clinical failures. Our BioCELL mitochondrial function assays provide a real-time energetics approach for probing mitochondrial energy metabolism in living cells using extracellular flux technology, improving the precision and sensitivity of in-vitro mitochondrial testing.
The importance of mitochondrial toxicity
Common toxicities linked with off-target drug-induced mitochondrial toxicity
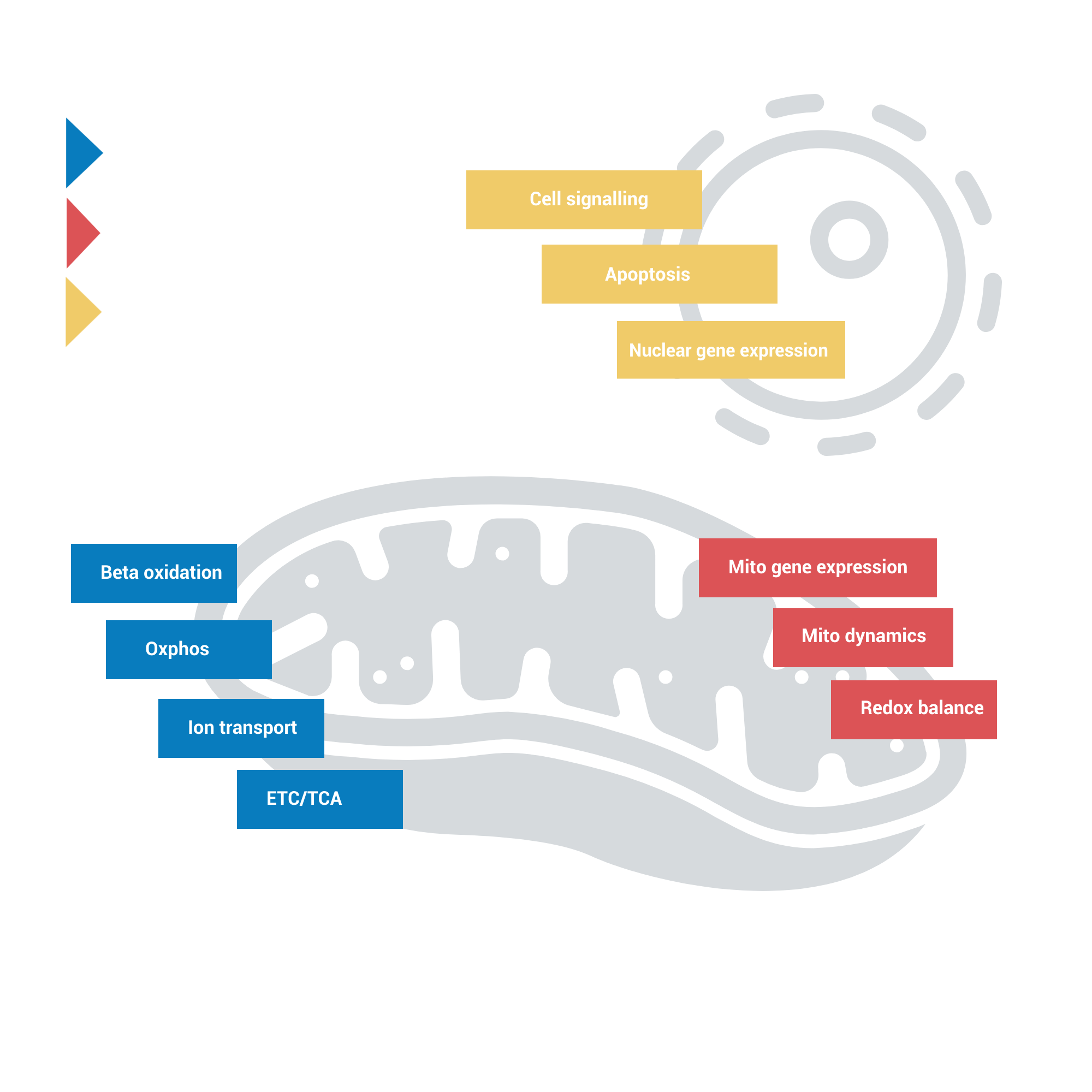
Mitochondria are central energy hubs in cells and pivotal for maintaining cellular homeostasis. Changes to the mitochondrial network are thus susceptible to off-target side effects of drugs. Given the many possible targets of drug-induced mitochondrial toxicity, understanding compound effects on mitochondrial energy metabolism in real-time using a sensitive assay such as our BioCELL MitoTOX assay provides a means to explore the magnitude of change in mitochondrial function and identify the mechanism of action as a result of drug-induced toxicity.
-
Hepatotoxicity

-
Cardiotoxicity

-
Neurotoxicity

-
Nephrotoxicity

-
Immunotoxicity


Since the late 1990s many drugs have been withdrawn from the market due to adverse effects on mitochondrial function. Some examples and their associated mitochondrial defect(s) are shown in the list below.
-
Therapeutic class: Antidiabetic
Toxicity: Hepatotoxicity
Defect(s): OxPHOS inhibition, mtDNA depletion and loss of membrane potential
Status: Withdrawn
-
Therapeutic class: Anxiolytic
Toxicity: Hepatotoxicity
Defect(s): OxPHOS inhibition and mPTP opening
Status: Withdrawn
-
Therapeutic class: NSAID
Toxicity: Cardiotoxicity
Defect(s): Respiratory complex IV inhibition, MMP and oxidative stress
Status: Withdrawn
-
Therapeutic class: Anticancer
Toxicity: Cardiotoxicity
Defect(s): Swelling/MMP
Status: Black box warning
-
Therapeutic class: Antiarrhythmic
Toxicity: Cardiotoxicity
Defect(s): OxPHOS inhibition and mPTP opening
Status: Black box warning
-
Therapeutic class: Antiretroviral
Toxicity: Neurotoxicity
Defect(s): Decreased mitochondrial function and mtDNA damage
Status: Withdrawn
-
Therapeutic class: NSAID
Toxicity: Hepatotoxicity and Nephrotoxicity
Defect(s): Inhibition of respiratory complex IV
Status: Withdrawn
De-risking your drug discovery pipeline
Our mitochondrial toxicity assays can be implemented at different times throughout your workflow to de-risk your drug discovery pipeline and ensure consumer safety.
Our mitochondrial toxicity assays
-
BioCELL mitoTOX
Probe real-time, direct measurements of mitochondrial metabolism with high sensitivity and specificity. Our solution for high-throughput compound screening.
-
BioCELL proFILE
An all-in-one solution to metabolically profile cells using real-time measurements of mitochondrial and glycolytic energy metabolism.
-
BioCELL adVANCED
Uncover mechanisms underlying compound effects on mitochondrial toxicity by directly measuring mitochondrial respiratory complex activity in real-time.